Alkamides
"In alkamides, amines are combined with unsaturated fatty acids by amide linkages, forming unbranched chains with one or more double and/or triple bonds." [Pengelly TCMP]
"The alkamides, also known as alkylamides, are fatty acid amides which vary in structure and function. Alkamides are found in nature in over 100 plant species, where they are thought to act as a defense against herbivory [1]. Alkamides contain a fatty acid tail, which can vary in the number of carbons and unsaturations, an amide group, and a variable headgroup." [Johnstone&Laster,2021]
"Alkamides are responsible for the sharp, burning or tingling taste associated with herbs and spices such as prickly ash bark (Zanthoxylum spp.), black pepper (Piper nigrum), Echinacea angustifolia, E. purpurea and cayenne (Capsicum spp.). Capsicum oleoresin contains several phenolic amides including capsaicin." [Pengelly TCMP]
"Alkamides are a large group of natural products occurring in at least 33 plant families. They possess a broad range of biological activities including analgesic, cannabimimetic, and immunomodulatory properties. Interestingly, two recent extensive reviews on alkamides did not list Ranunculaceae as an alkamide-producing family [108,109]." [Jetter FSAP]
As of this 2010 publication, it's reported that approximately 150 nitrogen-containing alkamides have been identified in higher plants. [Wink BBPSM]
Compounds and Sources
- Pellitorine: Piper longum [Johnstone&Laster,2021]
- pipercyclobutanamide A: Piper nigrum - long pepper [Root] [Huu, Duc Minh Nguyen, et al., 2021]
- Piperlongumine: Piper longum - long pepper [Root] [Tripathi&Biswal,2020]
- Sanshool: Zanthoxylum clava-herculis (α-sanshool) [Johnstone&Laster,2021]
- Spilanthol: Spilanthes sp., Spilanthes acmella [Johnstone&Laster,2021]
- undeca-2Z,4E-diene-8,10-diynic acid isobutylamide: Echinacea [Johnstone&Laster,2021]
- dodeca-2E,4E,8Z,10E/Z-tetraenoic acid isobutylamide: Echinacea [Johnstone&Laster,2021][Tracy HPTCP]
- dodeca-2E,4E-dienoic acid isobutylamide: Echinacea [Johnstone&Laster,2021][Tracy HPTCP]
- undeca-2E-ene-8,10-diynoic acid isobutylamide: Echinacea [Johnstone&Laster,2021]
- Undeca-2D/Z-ene-8,10-diynoic acid isobutylamides: Echinacea [Tracy HPTCP]
- Dodeca-2D,4Z-diene-8,10-diynoic acid isobutylamide: Echinacea [Tracy HPTCP]
- Dodeca-2E-ene-8,10-diynoic acid isobutylamide: Echinacea [Tracy HPTCP]
- Dodeca-2E,4E,8Z-trienoic acid isobutylamide: Echinacea [Tracy HPTCP]
Compound Details
Pellitorine: "pellitorine has also been shown to be an effective insecticidal agent against the housefly and Aedes aegypti mosquito [20, 21]." [Johnstone&Laster,2021]
pipercyclobutanamide A: "From the EtOH-soluble extract of the roots of Piper nigrum, one new dimeric alkamide, pipercyclobutanamide D (1) was isolated.... Compound 1 showed α-glucosidase inhibitory activity with an IC50 value of 158.5 µM. In addition, compound 1 exhibited cytotoxicity against the MCF-7 and HepG2 cell lines with the IC50 values of 45.6 and 63.9 µM, respectively." [Huu, Duc Minh Nguyen, et al., 2021]
Piperlongumine: "Piperlongumine (piplartine) is a well-known biologically active alkaloid/amide found in the roots of the plant Piper longum (long pepper) [7]." [Tripathi&Biswal,2020]
"Piperlongumine, a white to beige biologically active alkaloid/amide phytochemical, has high pharmacological relevance as an anticancer agent. Piperlongumine has several biological activities, including selective cytotoxicity against multiple cancer cells of different origins at a preclinical level." [Tripathi&Biswal,2020]Spilanthol: "Spilanthol is the predominant alkamide found in Spilanthes sp. with several other alkamides reported in lesser quantities [29]. Commercial preparations of spilanthol are as available for use as oral analgesics and to provide a long-lasting mint flavor in toothpastes [2].... Spilanthol displays structural similarities to capsaicin, the ligand for the nociceptor channel TRPV1, which may account for its analgesic properties [23]. Isolated spilanthol also displays immunomodulatory properties in vitro causing dose dependent reduction in macrophage activation and nitric oxide (NO) production, as well as inhibition of cytokine production and NF-κB activation [31]." [Johnstone&Laster,2021]
Cinnamides
"Cinnamides can be considered a subclass of alkamides in which the acid portion is derived from hydroxycinnamic acids such as caffeic, ferulic, or isoferulic acid, and the amine part is derived from amino acids or biogenic amines. The identification of black cohosh cinnamides, many of which are new natural products, was accomplished by a simple strategy that will be described here using the unique black cohosh cinnamides caffeoyl (46) and feruloyl/isoferuloyl arginine (47,48) as exemplary compounds." [Jetter FSAP]
Isobutylamides
"Isobutylamides are a subclass of alkamides based on the amine group 2-methylpropyl. They first aroused the interest of researchers for their insecticidal activities, being toxic to numerous classes of insects including the ubiquitous housefly and mosquito. Upon further investigation, it was obvious the most active insecticidal compounds were the ones that produced the most potent sialagogue (stimulating saliva flow) effects in humans (Brinker 1991/92). Isobutylamides so far investigated are derived from four plant families—Asteracea, Rutaceae, Piperaceae and Aristolochiaceae...." [Pengelly TCMP]
Capsaicinoids
"These compounds are responsible for the hotness in chillis (hot peppers), the degree of pungency being related to the length of the acid side chain. Total capsaicinoid content is around 1% of the dried fruit, the majority of which is usually capsaicin. Structurally, capsaicinoids are vanillyl-acyl amide analogues (Tucker and Debaggio 2000).
Capsaicinoids stimulate receptors (known as vanillinoid receptors) on cutaneous sensory neurons, resulting in a massive release of neuropeptides including so-called ‘substance P’ molecules responsible for pain transmission to the brain and modulation of local inflammatory responses. Topical applications of medications containing capsaicinoids deplete the neuropeptides, therefore preventing transmission of pain signals to the brain (Warber 1999). Hence the successful use of capsicum-based preparations for treatment of neuralgias, diabetic neuropathy and joint inflammation." [Pengelly TCMP]Pharmacology
"Alkamides can be readily detected by organoleptic means, by placing a very small quantity on the tongue. The initial sharp sensation and saliva production is followed by a local anaesthetic or numbing effect. Not surprisingly many are associated with management of toothache—Spilanthes and Zanthoxylum spp. are both known in their respective regions as ‘toothache plant’. The most significant actions of alkamides are analgesic, antiinflammatory, counter-irritant, sialagogue, vermifuge, digestive and circulatory stimulation. Other likely effects are spasmolytic, carminative and immune stimulation." [Pengelly TCMP]
"Similar to alkaloids, some alkamides from Piper spp. are reported to exert in vitro schistosomicidal activity. Piplartine (compound 12, Fig. 8-4), an amide found in several Piper spp., such as Piper tuberculatum, showed in vitro activity against adult S. mansoni worms. It was reported that treatment with 15.8 mM piplartine reduced motor activity and egg production, as well as killing all adult worms. Piplartine also induced morphologic changes in the tegument, causing extensive tegumental destruction and damage to the parasite tubercles. This damage was dose dependent in the range of 15.8-630.2 uM. More schistosomicidal and phytochemical studies should be conducted with Piper spp. since most have shown to be active against schistosomes [91]." [Rai FMDR]
Stability
"Alkamides are known to be subject to degradation over time. Studies on Echinacea purpurea alkamides showed that, while drying had no effect, most of the compounds were lost after the dried roots had been stored for a little over a year (Perry and van Klink 2000). Loss of alkamides was rather less when the roots were stored at subzero temperatures. Preservation in ethanolic tincture form is an effective method of retaining these compounds. Chopping the root produces only minimal losses." [Pengelly TCMP]
"A recent study has analyzed the effect of some processing variables on the content of solvent-soluble constituents. Chopping altered the level of some alkamides slightly in E. purpurea roots but drying had no significant effect on the amounts detected. Levels of all alkamides fell by over 80% on storage at room temperature for 16 months and also fell significantly even when the plant material was stored at 18oC (82)." [Packer HTM]
Echinacea Specifics
"Other constituents found in Echinacea include 15 lypophilic alkamides, most of which contain one or two acetylenic bonds." [Shahidi PIF]
Echinacea (Echinacea purpurea, Echinacea angustifolia, or Echinacea pallida): "The alkamides comprise a complex mixture of unsaturated fatty acids as amides with 2-methylpropanamine (isobutylamine) or 2-methylbutanamine, amines which are probably decarboxylation products from valine and isoleucine respectively. The acid portions are predominantly C11 and C12 diene-diynes or tetraenes (Figure 3.13). These compounds are found throughout the plant, though relative proportions of individual components vary considerably. The root of Echinacea purpurea contains at least 12 alkamides (about 0.6%), of which C12 diene-diynes predominate; levels of these compounds fall significantly during drying and storage." [MNP Dewick]
"More than 20 alkamides, mostly isobutylamides of C11–C16 straight-chain fatty acids with olefinic or acetylenic bonds, or both, are found in the roots; the highest concentration is in E. angustifolia, followed by E. purpurea, and the lowest is in E. pallida. The main alkamide is a mixture of isomeric dodeca-2,4,8,10-tetraenoic acid isobutylamides." [WHO SMPV.1]
"Hinz et al. (2007) showed that alkamides derived from E. angustifolia roots may contribute to the pharmacological action by inhibiting COX-2-dependent PGE2 formation at sites of inflammation (Hinz et al., 2007)." [Seeram HM]
"Several alkamides from E. purpurea showed ca. 48% and 30% inhibition of COX-1 and -2, respectively, at 100 mg/mL.20" [Leung ECNI]
"Polyunsaturated alkamides isolated from Echinacea angustifolia inhibit cyclooxygenase and 5-lipoxygenase in vitro (Mullr-Jakic et al., 1994)." [TCPH]
"Preclinical studies indicate that Echinacea constituents modulate immune mechanisms and there is an increasing evidence that lipophilic Echinacea preparations containing alkamides can suppress stress-related cellular immune responses.... Chicca et al. reported that a synergism between Echinacea lipids (mixtures of alkamides) from radix and herba tinctures are likely to mediate superior anti-inflammatory and immuno-pharmacological effects over single compounds." [Seeram HM]
Antiviral: "Recently, as an alternative approach to the common anti-viral therapy (vaccines and a few anti-viral drugs), a commercial standardized extract of Echinacea (Echinaforce®, an ethanolic extract of herb and roots of E. purpurea) has been studied in order to elucidate the nature of its anti-influenza virus activity. Human H1N1-type IV, highly pathogenic avian IV (HPAIV) of the H5- and H7-types, as well as swine origin IV (S-OIV, H1N1), were all inactivated in cell culture assays by the E. purpurea preparation at concentrations ranging from the recommended dose for oral consumption to several orders of magnitude lower (MIC100 values increased from 0.32 µg/mL Echinaforce® for 102 PFU/mL virus, up to 7.5 µg/mL Echinaforce® for 105 PFU/mL). Hemagglutination assays showed that the extract inhibited the receptor binding activity of the virus, suggesting that the Echinacea extract interferes with the viral entry into cells. Furthermore, a Tamiflu®-resistant virus was just as susceptible to E. purpurea preparation as the wild-type virus (Pleschka et al., 2009). Another approach for an anti-viral action has been investigated with herpes simplex virus type-1 (HSV-1), which often causes recurrent ocular disease during the latency period. An Echinacea polysaccharide fraction (isolated from E. purpurea aerial parts at a concentration of 20 mg/mL; Zardband Pharmaceutical Company) led to a reduced latency rate and exerted an anti-viral action on the development of recurrent HSV-1 disease when supplied prior to infection (Ghaemi et al., 2009)." [Seeram HM]
"Recently, Echinacea alkamides have been reported to be a new class of cannabinomimetics modulating tumor necrosis factor α gene expression via the cannabinoid type 2 receptor" [Seeram HM]
"Overall, the studies have shown that most of the alkamides present in Echinacea preparations are bioavailable from an oral dose of different Echinacea formulations...." [Seeram HM]
"So far, the therapeutic activity of Echinacea cannot be unambiguously attributed to any particular constituent. However, recent pharmacological data suggest that within the active principle of Echinacea, the alkamides showed the most abundant properties with marked immunomodulatory, anti-inflammatory, anti-viral, and bactericidal effects.... According to these results and the CB receptor binding activities, and since bioavailability has been demonstrated, alkamides seem to be clinically highly relevant constituents of Echinacea (Woelkart and Bauer, 2007)." [Seeram HM]
Insecticide: "Different alkamides of E. purpurea produced 100% mortality in Aedes aegyptii mosquito larvae when tested in vitro at 100 mg/mL.20" [Leung ECNI]
Misc Alkamides
Artemisia dracunculus: "The aerial parts contain the alkamides, pellitorine, and neopellitorine A and B. The latter two were isolated as new compounds from A. dracunculus.3" [Leung ECNI]
Piper nigrum: "Aqueous and organic extracts and many alkamides of black pepper exhibited antibacterial activity against both Gram-positive and Gram-negative bacteria.14,30 Larvicidal and insecticidal activities have also been reported.7,9,10,13,31,32" [Leung ECNI]
"As detailed above, many alkamides are present in E. angustifolia roots where they are particularly abundant in quantity. Their structures are derived from undeca- and dodecanoic acid and differ from one another in the degree of unsaturation and the configuration of the double bonds (66). In E. purpurea roots there are characteristic differences in that most of the 11 alkamides possess a 2,4-diene moiety. This makes a convenient marker for distinguishing the source of the material (4,75). The aerial parts contain similar constituents (67,68). The concentration of most of these compounds is significantly less than 1% (0.001–0.151%). Alkamide levels in various parts of E. purpurea differ sufficiently to suggest that measurements of their distribution could be used to determine the origin of extracts (79). The photochemical instability of such compounds would require considerable caution in sample preparation and handling (80)." [Packer HTM]
Phyllanthus niruri: "Two alkamides, E,E-2,4-octadienamide and E,Z-2,4-decadienamide have also been isolated from the plant with anti-plasmodial activities. The anti-plasmodial activity has been attributed to the presence of α, β, γ, δ-unsaturated conjugated amide (Sittie et al. 1998)." [Rai FMDR]
References
- Huu, Duc Minh Nguyen, et al., 2021 - Huu, Duc Minh Nguyen, et al. "Pipercyclobutanamide D, a new member of the cyclobutanamide-type alkaloid, from the roots of Piper nigrum." Journal of Asian Natural Products Research 23.9 (2021): 906-912.
- Johnstone&Laster,2021 - E. Johnstone, Stephanie, and Scott M. Laster. ‘The Structure and Function of Alkamides in Mammalian Systems’. Natural Drugs from Plants, IntechOpen, 11 May 2022. Crossref, doi:10.5772/intechopen.98198.
- Tripathi&Biswal,2020 - Tripathi, Surya Kant, and Bijesh Kumar Biswal. "Piperlongumine, a potent anticancer phytotherapeutic: Perspectives on contemporary status and future possibilities as an anticancer agent." Pharmacological Research 156 (2020): 104772.
Amines
"Amines are derivatives of ammonia in which one or more of the hydrogen atoms have been replaced by an alkyl or aryl group, so like ammonia they are basic compounds." [X Ge et al.,2011]
"Amines are emitted by a wide range of sources and are ubiquitous atmospheric organic bases. Approximately 150 amines and about 30 amino acids have been identified in the atmosphere." [X Ge et al.,2011]
"Biologically active amines are normal constituents of many foods and have been found in cheese; sauerkraut; wine; and putrid, aged, or fermented meats. These low molecular weight organic bases do not represent any hazard to individuals unless large quantities are ingested or natural mechanisms for their catabolism are inhibited or genetically deficient." [Rice et al., 1976]
"Biogenic amines are conveniently divided into aliphatic monoamines, aliphatic di- and polyamines and aromatic amines. These compounds are shown to fulfill an array of roles in cellular metabolism. Thus, amines are needed for growth and development and their metabolism appears to be coordinated with the cell cycle." [Bouchereau et al., 2000]
"Biogenic amines (BA) are organic compounds commonly found in food, plants and animals, as well as microorganisms that are attributed with the production of BAs. They are formed as an effect of a chemical process: the decarboxylation of amino acids. Factors determining the formation of BAs include the availability of free amino acids and the presence of microorganisms that show activity with respect to carrying out the decarboxylation process. On the one hand, BAs are compounds that are crucial for maintaining cell viability, as well as the proper course of the organism's metabolic processes, such as protein synthesis, hormone synthesis and DNA replication. On the other hand, despite their positive effects on the functioning of the organism, an excessive content of BAs proves to be toxic (diarrhea, food poisoning, vomiting, sweating or tachycardia). Moreover, they can accelerate carcinogenesis. " [Wójcik et al., 2020]
Polyamines
"Polyamines are found in foods of both animal and plant origin, either in a free or conjugated form. Conjugated polyamines are found in plant-derived foods mainly linked to phenolic compounds (4, 24). In foods, spermidine and spermine are primarily naturally present, coming from raw plant and animal tissues, whereas putrescine may also be formed by the activity of fermentative or contaminating microorganisms (12, 53). It has also been described that spermidine and spermine may partly have a bacterial origin, especially in fermented products (12, 54, 55). Therefore, processing and storage conditions can influence the total content of polyamines." [Muñoz-Esparza, Nelly C., et al.,2019]
"The polyamines spermine, spermidine, and putrescine are involved in various biological processes, notably in cell proliferation and differentiation, and also have antioxidant properties. Dietary polyamines have important implications in human health, mainly in the intestinal maturation and in the differentiation and development of immune system. The antioxidant and anti-inflammatory effect of polyamine can also play an important role in the prevention of chronic diseases such as cardiovascular diseases. In addition to endogenous synthesis, food is an important source of polyamines. Although there are no recommendations for polyamine daily intake, it is known that in stages of rapid cell growth (i.e., in the neonatal period), polyamine requirements are high. Additionally, de novo synthesis of polyamines tends to decrease with age, which is why their dietary sources acquire a greater importance in an aging population." [Muñoz-Esparza, Nelly C., et al.,2019]
Effects of Culinary Treatment
"Culinary treatment can potentially decrease the polyamine content in foods by two possible mechanisms: (a) transfer to the cooking water or (b) due to the high temperatures reached in some types of cooking. The few studies evaluating the effect of culinary treatment on polyamines report variable results, depending on the type of cooking and the food studied. Polyamine contents after the boiling of certain vegetables (spinach, cauliflower, and potatoes) were significantly reduced by transfer to the cooking water, especially putrescine, as this is the most water-soluble polyamine. However, the same cooking process did not induce losses in other types of food (peppers, peas, and asparagus) (84). Another study found no significant differences in polyamine levels between raw and boiled vegetables (carrots, broccoli, cauliflower, and potatoes), although the low number of samples analyzed (two per food type) was a limiting factor (9). In meat subjected to a cooking process involving a large amount of water (stewing and boiling), no significant losses of spermidine and spermine were observed either (23, 53). In the case of some cooking techniques that involved higher temperatures (53) described that roasting, grilling, or frying produced losses of up to 60% of spermidine and spermine in chicken meat." [Muñoz-Esparza, Nelly C., et al.,2019]
Common Amines
- tyramine
- histamine
- phenethylamine
- putrescine
- spermidine
- spermine
- phenolamines
- catecholamines
- imidazolamines
Indolamines
Melatonin: "Melatonin is an amphipathic indolamine molecule ubiquitously present in all organisms ranging from cyanobacteria to humans. The pineal gland is the site of melatonin synthesis and secretion under the influence of the retinohypothalamic tract. Some extrapineal tissues (skin, lens, gastrointestinal tract, testis, ovary, lymphocytes, and astrocytes) also enable to produce melatonin. Physiologically, melatonin regulates various functions like circadian rhythm, sleep–wake cycle, gonadal activity, redox homeostasis, neuroprotection, immune-modulation, and anticancer effects in the body. Inappropriate melatonin secretion advances the aging process, tumorigenesis, visceral adiposity, etc." [Samanta, Saptadip.,2020]
"Using the described method of extraction and after correction of the values for the recovery rate (about 40%), the calculated concentrations of melatonin in walnuts was 3.5 +- 1.0 ng/g." [Reiter et al.,2005]
Serotonin: [Text missing in source]
Methylamines
[Text missing in source]
Amine Compounds and Sources
- Melatonin (Indolamine): "Melatonin has been reported in edible fruits (grape, banana, strawberries, apple, pineapple, Kiwi fruit, tart cherries, etc.), vegetables (tomato, pepper, mushroom, cabbage, cauliflower, cucumber, carrot, beetroot, etc.), nuts and raw seeds (walnuts, sunflower, green cardamom, fenugreek, white and black mustard, etc.), juices and beverages (wines, green and roasted coffee beans, decoction brew)." [Ramakrishna et al.,2021]
Amines & Alkamides
Group: Nitrogenous Compounds
Google Gemini - Created with AI prompt, using existing notes (This page may require extra scrutiny)
Overview & Biosynthesis
Amines are derivatives of ammonia in which one or more hydrogen atoms are replaced by alkyl or aryl groups. They are ubiquitous atmospheric bases emitted by a wide range of sources.(1) In biological systems, they are often formed via the decarboxylation of amino acids.(2)
Alkamides (alkylamides) are a specialized subclass where amines combine with unsaturated fatty acids via amide linkages. They are distinct lipophilic compounds found in over 33 plant families, including Asteraceae and Piperaceae.[Pengelly TCMP], (3) Unlike simple amines, alkamides often function as plant defense mechanisms against herbivory and possess significant pharmacological activity in humans, including immunomodulation and local anesthesia.(4), [Pengelly TCMP]
Alkamides
Fatty Acid AmidesResponsible for the "tingling" or numbing sensation (paresthesia) in herbs like Echinacea and Zanthoxylum. They are potent sialagogues (saliva promoters) and immunomodulators.[Pengelly TCMP]
- Mechanism: Spilanthol and similar alkamides display structural similarities to capsaicin, acting on nociceptor channel TRPV1.(4)
- Local Sources: Achillea millefolium (Yarrow) roots contain pellitorine (deca-2E,4E-dienoic acid isobutylamide) and other olefinic acid amides.(5)
- Stability: Levels fall by over 80% if stored at room temperature for 16 months. Ethanolic tincture is the most effective preservation method.[Pengelly TCMP], (6)
| Compound | Source |
|---|---|
| Spilanthol | Spilanthes acmella (Toothache plant)(4) |
| Pellitorine | Achillea millefolium, Piper nigrum(5) |
| Piperlongumine | Piper longum (Long Pepper)(7) |
| Undeca-2E-ene-8,10-diynoic acid | Echinacea angustifolia(6) |
Biogenic Amines
Polyamines & IndolaminesLow molecular weight organic bases involved in growth and development.
| Class | Notes |
|---|---|
| Polyamines (Spermine, Spermidine) |
Found in cheese, sauerkraut, and fermented meats. Essential for cell proliferation but toxic in excess.(8) High heat (frying/grilling) can degrade them by up to 60%.(8) |
| Indolamines (Melatonin, Serotonin) |
Melatonin is present in walnuts (3.5 ng/g), cherries, and tomatoes.(9) It acts as an antioxidant and regulates circadian rhythms.(10) |
| Methylamines (Trimethylamine) |
In plants like Tea (Camellia sinensis), methylamine is oxidized to formaldehyde, serving as a precursor for purine alkaloids like caffeine and theobromine.(11) |
Clinical Significance & Hazards
Toxicity (Amines): Polyamines are crucial for maintaining cell viability, yet excessive content in food sources can prove to be toxic.(2)
Pharmacology (Alkamides):
- Immune: Alkamides in Echinacea bind to Cannabinoid (CB2) receptors, modulating TNF-α and suppressing stress-related immune responses.(12), (13)
- Antiviral: Echinacea ethanolic extracts inhibit receptor binding of Influenza A (H1N1, H5, H7), interfering with viral entry.(13)
- Insecticide: Pellitorine and isobutylamides are toxic to houseflies and mosquito larvae (Aedes aegypti).(4)
Figure 1: Accurate Chemical Structure and Biological Mechanism of Spilanthol
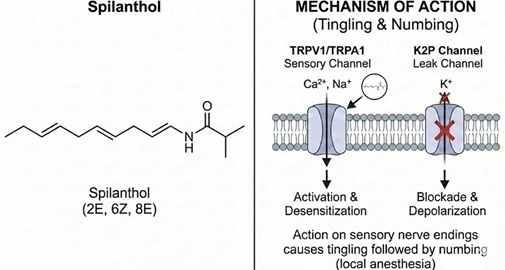
This diagram provides a precise representation of (2E, 6Z, 8E)-N-isobutyldeca-2,6,8-trienamide, the primary pungent component of Acmella oleracea. The structure correctly illustrates the specific cis-trans geometry of the triene chain. The right panel effectively maps the dual mechanism of action: activation of TRP sensory channels (responsible for the tingling "electric" sensation) and the inhibition of K2P (potassium leak) channels, which leads to depolarization and the localized numbing effect used in traditional medicine for toothaches.
Figure 2: Biochemical Analysis: Valine Decarboxylation Pathway
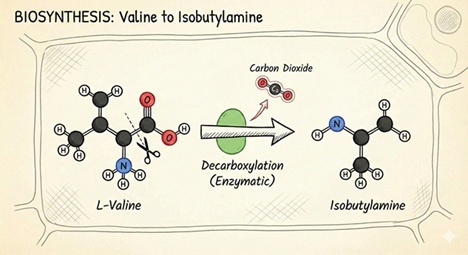
This illustration depicts the biosynthetic transition from a primary amino acid to a biogenic amine precursor, a critical step for the synthesis of secondary metabolites like the alkamides found in Echinacea.
Original Notes
Detailed Compound Data & References
Alkamides Inventory
Reported ~150 nitrogen-containing alkamides in higher plants.(14)
- Pipercyclobutanamide A: Isolated from Piper nigrum roots; shows cytotoxicity against MCF-7 cell lines.(15)
- Piperlongumine: Anticancer agent from Piper longum; selective cytotoxicity against multiple cancer cells.(7)
- Sanshool: α-sanshool found in Zanthoxylum clava-herculis (Prickly Ash).(4)
References
- (1) Ge, X. et al. (2011). "Atmospheric amines–Part I. A review." Atmospheric Environment 45(3).
- (2) Wójcik, W. et al. (2020). "Biogenic amines: formation, action and toxicity." Journal of the Science of Food and Agriculture 101.
- (3) Jetter. "Function and Synthesis of Alkamides in Plants." (FSAP).
- (4) Johnstone, E. & Laster, S.M. (2021). "The Structure and Function of Alkamides in Mammalian Systems." Natural Drugs from Plants.
- (5) Veryser, L. et al. (2017). "N-alkylamide profiling of Achillea ptarmica and Achillea millefolium extracts." Journal of Pharmaceutical Analysis 7(1): 34-47.
- (6) Packer, L. "Herbal Medicine: Biomolecular and Clinical Aspects." (HTM).
- (7) Tripathi & Biswal (2020). "Piperlongumine, a potent anticancer phytotherapeutic." Pharmacological Research 156.
- (8) Muñoz-Esparza, N.C. et al. (2019). "Polyamines in food." Frontiers in Nutrition 6.
- (9) Reiter, R.J. et al. (2005). "Melatonin in walnuts..." Nutrition 21: 920–924.
- (10) Samanta, S. (2020). "Melatonin: an endogenous miraculous indolamine..." J Cancer Res Clin Oncol 146.
- (11) Suzuki, T. (1973). "Metabolism of methylamine in the tea plant (Thea sinensis L.)." Biochemical Journal 132(4): 753-763.
- (12) Chicca et al. Cited in Seeram, "Herbal Medicines" (HM).
- (13) Seeram, N. "Herbal Medicines: Biomolecular and Clinical Aspects."
- (14) Wink, M. "Biochemistry of Plant Secondary Metabolism." (BBPSM).
- (15) Huu, Duc Minh Nguyen, et al. (2021). "Pipercyclobutanamide D..." Journal of Asian Natural Products Research 23.9.