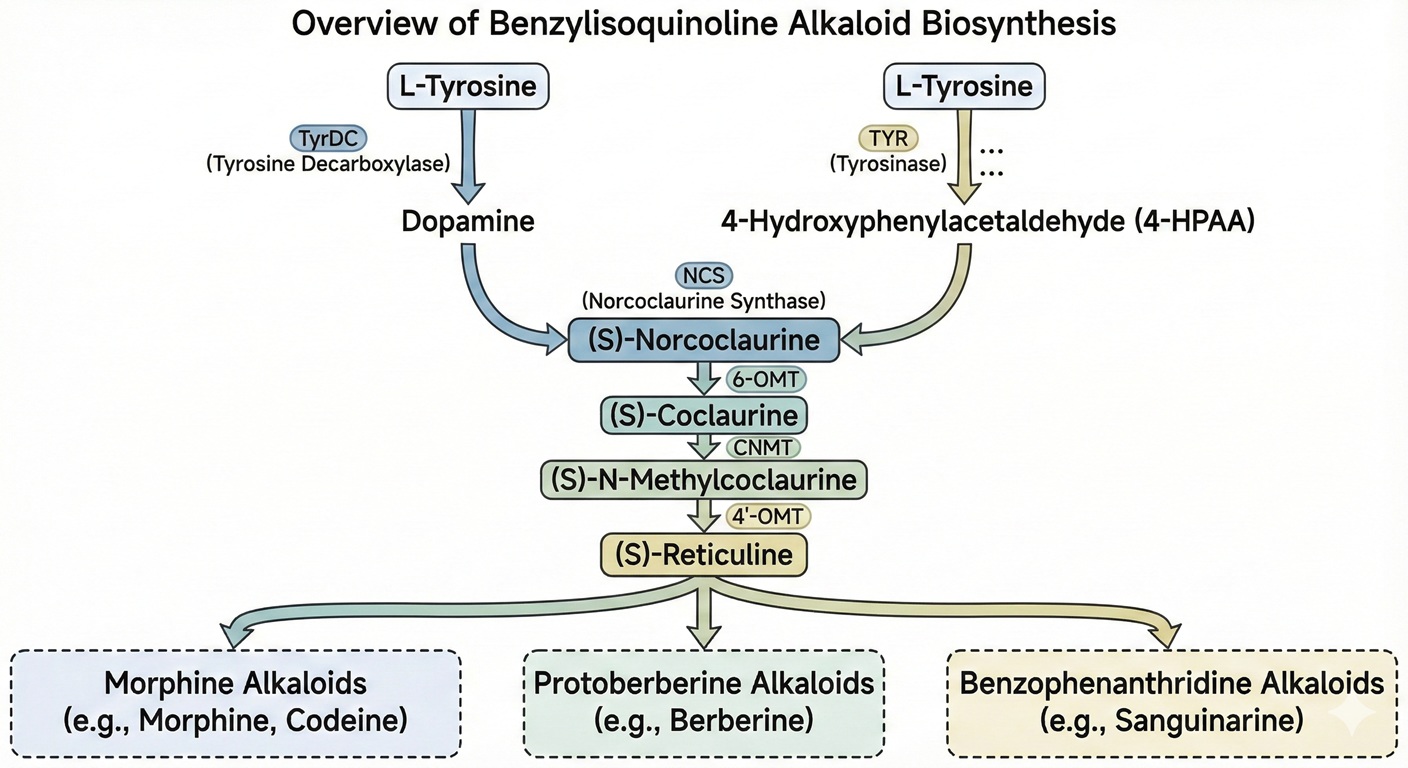
Berberine (Isoquinoline)
- Berberine is from the protoberberine group of isoquinoline alkaloids [Ramawat NP]
- "Pharmacological data also suggest that combining berberine-containing herbs with other botanical synergists might prove even more effective." (CBMed)
- Berberine salts have been used in ophthalmic products, usually in eye drops and eyewashes. [Leung ENCI]
- Interactions:
- Ciclosporin: "Berberine appears to increase the bioavailability and trough blood levels of ciclosporin.... Although the increase in ciclosporin levels is not sufficiently severe to suggest that the concurrent use of berberine should be avoided, it may make ciclosporin levels less stable." [HMI Stockey]
- Paclitaxel: An in vitro study found that pre-treatment with berberine blocked the anticancer effects of paclitaxel in six cancer cell line cultures (oral cancer, gastric cancer and colon cancer). [HMI Stockey]
Distribution: Mahonia spp. (Oregon Grape), Berberis spp., Coptis spp., Hydrastis canadensis.
Activities: Antibacterial, antifungal, amoebicidal, antiarrhythmic, antiinflammatory. "Berberine has marked antibacterial effects and is used as a bitter tonic... Since it is not appreciably absorbed by the body, it is used orally in the treatment of various enteric infections." (PFAF)